
Report Adverse Reaction

Approved fees Schedules

Products Recall And Alert

For Stakeholders

Submit a Complaint

FAQs

Submit Advert Application

Submit Advert Application

Consumer Education
News & Events
Latest News
2024-08-12 00:00:00
ONLINE PURCHASE OF MEDICAL DEVICES FOR PERSONAL USE2024-04-17 00:00:00
Ghana's COVID-19 Safety Monitoring Effects Receive Global Acclaim2024-04-05 00:00:00
FDA AND AFROCET MONTGOMERY MEETS STAKEHOLDERS2024-03-13 00:00:00
NEW FDA FEES AND CHARGES EFFECTIVE MONDAY 18th MARCH 20242024-02-01 00:00:00
DRAFT GUIDELINE FOR PUBLIC CONSULTATION "FDA GUIDELINE ON IDENTIFICATION AND DATA CAPTURE FOR PHARMACEUTICAL TRACEABILITY"2023-11-17 00:00:00
JOHN OWUSU GYAPONG IS THE NEXT ARUA SECRETARY-GENERAL2023-11-17 00:00:00
FDA LAUNCHES THE NATIONAL TOBACCO CONTROL STRATEGYSAFETYWATCH UPDATE No. 04 - COVID-19 VACCINE SAFETY MONITORING WEEKLY UPDATE(23rd March to 29th March 2021)
Summary
This Update No. 4 provides the safety overview of the COVIID-19 vaccine in Ghana for the period 23rd to 29th March 2021. Since publication of Update No. 03 (16th-22nd March 2021) which provided safety overview for up to the third week of the vaccine deployment, an additional 47,894 doses of the Covishield Vaccine have been given with 106 persons reporting adverse events (commonly known as side effects) which are mostly mild flu-like symptoms; headache, fever, weakness, body ache and chills.
The Covishield Vaccine continues to be safe and its benefits outweigh possible risks. The reported side effects experienced after vaccination usually resolve within a day or two. Cumulatively, the reporting rate is about 3 adverse event reports for every 1000 persons vaccinated, which signifies that the FDA has in place, a well-established/robust safety monitoring system.
Highlights
- After 4 weeks of vaccinations, a total of 522,313 doses of Covishield Vaccine have been given with 1,733 persons reporting adverse events following immunization (AEFIs); this gives a reporting rate of about 3 reports per 1,000 doses administered.
- Additional 3 serious AEFI reports were received in the third week bringing the cumulate number of serious AEFIs to 15.
- The Joint COVID-19 Vaccine Safety Review (JCVSR) Committee had its third meeting on Friday, 26th March 2021 and perform causality assessment for five of the serious AEFI reports for which investigations were completed. The Committee determined that three of the serious events were coincidental (i.e. the AEFI that is caused by something other than the vaccine product, immunization error or immunization anxiety). The remaining two cases were classified as having consistent temporal relationship between vaccine administration and onset of the event, however there was insufficient definitive evidence for vaccine causing event hence monitoring of such cases should continue.
- In addition to the causality assessment of the adverse events, the JCVSR Committee also endorsed the FDA's continuous education and awareness creation programmes targeted at healthcare professionals and the public to encourage reporting of all suspected adverse events after COVID-19 vaccination.
Demographic Characteristics of Persons Who Reported Adverse Events
- Report by gender and age
The details on those who reported AEFIs were:
o Females - 957(55.2%)
o Males - 775 (44.7%)
o Unknown - 1 (0.1%)
o Mean age - 41 (SD =14.3)
Adverse Events Report by Monitoring Type
- Enhanced spontaneous reporting - 1338 (77.2%)
- Cohort event monitoring - 395 (22.8%)
Description of Adverse Events
The top ten most commonly reported adverse events based on the number of times these were reported during the four weeks of vaccinations is shown in Figure 1:
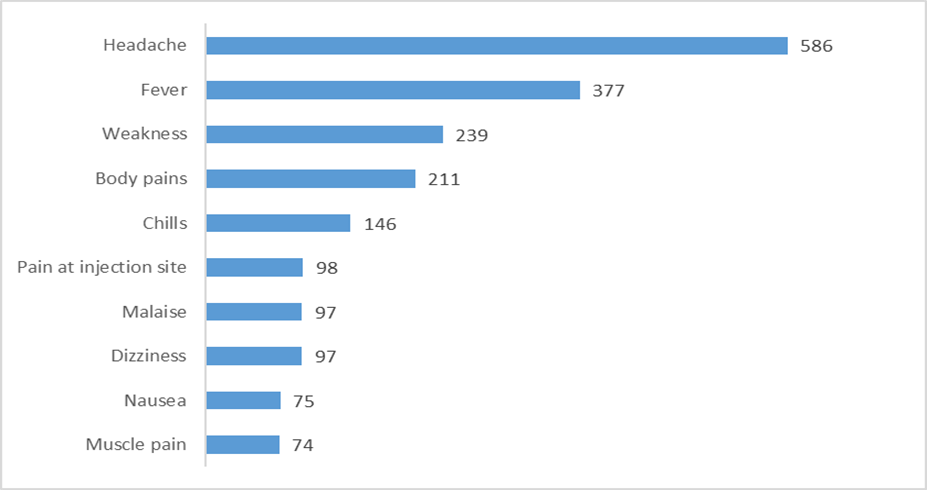
Fig. 1: The top ten most commonly reported events during the four weeks of vaccinations
Assessment of Adverse Event Reports
There might be no relationship between the AEFIs and the vaccine - it may be a coincidence that the adverse events occurred when the vaccine was given.
All adverse event reports received by the FDA are reviewed by the JCVSR Committee to find out the possible link between the events and the vaccine.
How to Report Adverse Events to COVID-19 Vaccines to the FDA
Reporting AEFIs help the FDA have more details about the safety of vaccines to enable any needed regulatory action to be taken to ensure public health and safety.
For any vaccine safety related information or to report AEFIs to COVID-19 vaccines, please contact the FDA through the following:
Mobile: 0244310297
Email: drug.safety@fda.gov.gh
Hotlines:0551112224/0551112225
Online: http://adr.fdaghana.gov.gh/patient.php
WhatsApp: 0551112225
Medsafety App: Download from the Apple store or Google play store
THE FDA MISSION
The FDA exist to ensure the safety, quality and efficacy of human and veterinary drugs, food, biological products, cosmetics, medical devices, household chemical substances and clinical trials, and the control of tobacco products through the enforcement of relevant standards to protect public health.

Subscription Management Centre












