Monday, 7th July 2025
The Food and Drugs Authority (FDA) has been alerted through the WHO Rapid Alert System for Falsified and Substandard Medical Products that the underlisted batches of the registered product; PARADENK SUPPOSITORIES (Paracetamol) with defective packaging materials have been imported and distributed in Ghana:
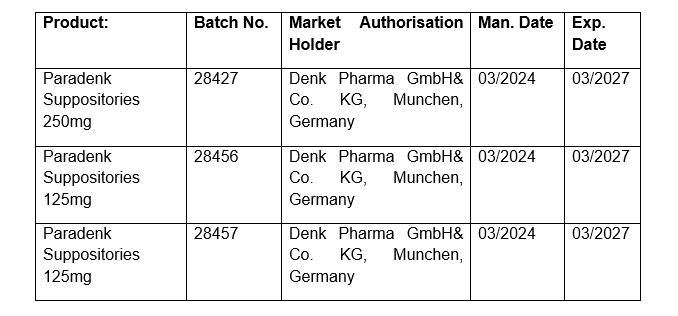
According to the alert, post–release quality control checks revealed defective packaging materials in the above mentioned batches of the pharmaceutical product. The damaged packaging materials do not guarantee safety and efficacy of the product until end of shelf-life due to lack of blister tightness.
PARADENK SUPPOSITORIES (Paracetamol) is used to relieve mild to moderate pain in children and in patients who have difficulty in swallowing medication. However, the safety and efficacy of medication can be compromised if packaging materials are defective. The marketing authorisation holder; Denk Pharma GmbH& Co. KG, Munchen, Germany has initiated a voluntary recall and the FDA is collaborating with the local agent, Denk Pharma in Ghana to ensure that the defective product batches are remove from the distribution chain.
Advise to the Public and Healthcare Professionals:
The general public and healthcare professionals are advised not to panic as the affected batches do not pose any immediate health risk to patients. If you have in stock or you are using any of the affected batch of PARADENK SUPPOSITORIES (Paracetamol), stop using it and submit the remaining stock to the nearest FDA office. You can call the FDA Safety Monitoring Department on 0244310297.
The FDA remains committed to ensuring health and safety of the public and is counting on the public and healthcare professionals to remove the affected batches of product from trade.


